Search
Additional Links Contact Us- Phone: +1 (800) 811-6990
- Toll-Free: +1 (800) 811-6990
- Europe: (514) 337-6990
- Fax: +1 (514) 335-0801
- Email: [email protected]
NEW! BRONCO® 4 Labeler ... Click Here

ProMach is your partner from start to finish. Our product brands are grouped into distinct business lines that make the most sense to our customers, covering every function of the production line: Filling, Bottling & Capping, Decorative Labeling, Flexibles & Trays, Pharma, Handling & Sterilizing, Labeling & Coding, Robotics & End of Line, and Systems & Integration.
learn moreThe AMS Series pharma sterilizers are pharmaceutical-grade autoclaves engineered for terminal sterilization using advanced steam-air mixture technology. Designed specifically for treating pharma-grade materials and products that require rapid drying and immediate readiness for further handling, this sterilization method protects pressure and heat-sensitive materials from damage.
Optimal air ventilation ensures uniform temperature distribution throughout the sterilizing chamber during all process phases. This makes the AMS Series ideal for sterilizing liquids in sealed or heat-sensitive containers, including:
Flexibility and versatility define the AMS Series, with devices available in various chamber sizes—ranging from 830 to 25,000 liters (29 to 883 cu ft). The chambers feature horizontal sliding double doors controlled by an electromechanical device, ensuring smooth operation. A silicon, FDA-compliant, swallowtail gasket—pneumatically pressurized by compressed air—guarantees door tightness.
The AMS Series pharma sterilizers are designed, manufactured, validated, and documented in accordance with the latest global standards, facilitating compliance with current Good Manufacturing Practices (cGMPs). These sterilizers provide the pharmaceutical industry with a reliable solution for handling sensitive products while maintaining efficiency and quality throughout the sterilization process.
Steelco pharma grade machines comply with the applicable requirements of the following regulations and industry accepted guidelines:
Chamber volume
from 830 litres / 29 cu ft
to 25.000 litres / 883 cu ft
Custom dimensions available upon request.
Overall dimensions
The overall dimensions may vary according to machine configuration and customer requirements. An electrical cabinet may be installed remotely from the machine as well as a steam generator depending on space and operational requirements.
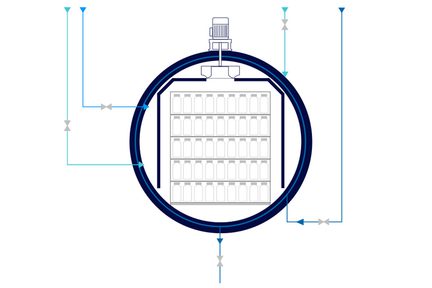
A high-quality AISI 316 L stainless steel chamber guarantee long-term reliability and steam cleanliness. Chamber is provided with a jacket for fast and uniform heating and cooling of the load or a heat exchanger inside the chamber. The machine body is insulated with ceramic material, and external cladding stainless steel–piping is in compliance with ASME BPE made of AiSI 316L stainless steel with tri-clamp fittings. FDA components in contact with the product are of sanitary type - a 3.1 certificate can be provided.

Pure steam is used in the chamber for heating liquids. Over-pressure filtered air is injected into the chamber to compensate for the rising temperature and pressure inside the products. Internal fans ensure a uniform temperature distribution, between 110°C to 135°C, to avoid cold spots across the chamber. Rapid heating and cooling are standard phases. The treated items are completely dry at the end of the cycle.
Steelco has evolved its technologies to integrate its devices among themselves and into existing automation systems and, at the software level, to integrate naturally with the client's software and customer management systems. We can install new equipment into an existing automation system by adopting a simple (HMI) or more sophisticated (SCADA) man-machine interface, with complete customization of access levels available. In compliance with the individual internal policies of each customer, Steelco offers remote service access by using virtual private networks to ensure the confidentiality and safety of all transferred data. Customized automation solutions that that are wholly integrated with the client's IS system and comprehensive network services such as integration with MES can be provided.
