Search
Additional Links Contact Us- Phone: +1 (800) 811-6990
- Toll-Free: +1 (800) 811-6990
- Europe: (514) 337-6990
- Fax: +1 (514) 335-0801
- Email: [email protected]
NEW! BRONCO® 4 Labeler ... Click Here

ProMach is your partner from start to finish. Our product brands are grouped into distinct business lines that make the most sense to our customers, covering every function of the production line: Filling, Bottling & Capping, Decorative Labeling, Flexibles & Trays, Pharma, Handling & Sterilizing, Labeling & Coding, Robotics & End of Line, and Systems & Integration.
learn moreThanks to a long experience within the pharmaceutical, nutraceutical, cosmetic, and food industries, Steelco offers a wide range of hot air static dryers to meet the most challenging market requirements.
The EG Series is designed for optimizing process parameters for effective, uniform, and gentle drying by hot air of materials such as granulates, pellets, powders, porous load, and equipment used in pharmaceutical industry processes. In addition, upon request and for particular types of applications – products containing a high percentage of alcohol, solvents, or materials classified as inflammable and potentially explosive – the electrical system and components of the machine can be supplied to be compliant with the flameproof type (ATEX Directives).
Available in single or double door pass-through hinged door versions, the devices feature chamber volumes from 330 to 10.000 liters (11.7 to 353 cu ft). On request and previous Steelco approval shall be possible to require customized chamber dimensions.
The EG Series of hot drying ovens are high-performing, reliable, and easy to use. Its cycle has been specifically designed to minimize time and energy consumption while maximizing efficiency and throughput-drying process temperature ranges from 50 to 160°C. In addition, upon request, the unit can be supplied with a self-washing system for performing CIP (Clean-In-Place) of the chamber and air ducts.
The EG Series is designed, manufactured, validated, and documented according to the latest global practices and standards to facilitate customers’ compliance with current Good Manufacturing Practices (cGMPs).
Steelco pharma grade machines comply with the applicable requirements of the following regulations and industry accepted guidelines:
Chamber volume
from 330 litres / 11.7 cu ft
to 10.000 litres / 353 cu ft
Custom dimensions available up on request
Overall dimensions
The overall dimensions may vary according to machine configuration and customer’s requirements. Electrical cabinet may be installed remotely from the machine depending on space and operational requirements.
The unit has electrical heating elements and air recycling fan(s) positioned on top of the chamber. Pre-filters and HEPA filters are installed on the inlet and exhaust air ducts to prevent cross-contamination. Monitoring of chamber pressure and HEPA filters efficiency by differential pressure transmitters. Absolute filters' housing provided with accessories necessary for the DOP test. The air cooling is carried out by a heat exchanger running by chilled or softened water.
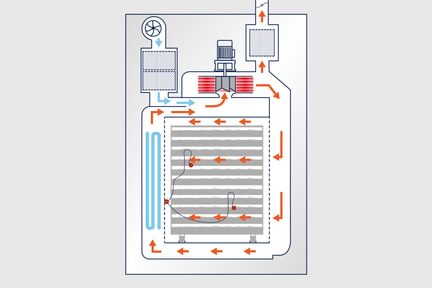
Differentially perforated walls, airspeed, and temperature control ensure an extremely air drying quality process. Tri-clamp connections for air quality monitoring and validation activities installed on fascia. As standard, one temperature product probe in chamber is provided for temperature monitoring. Upon request, additional temperature probes are provided.
